On February 20, 2026, the UK's Department of Health and Social Care announced that the PATHWAYS clinical trial into puberty blockers had been paused. The Medicines and Healthcare products Regulatory Agency (MHRA) raised "new concerns" about participant wellbeing and recommended a minimum participant age of 14, citing "unquantified risk" of long-term biological harms.
If you only caught the headlines that day, you were missing five years of context.
This is exactly the kind of story that breaks most monitoring tools. It spans multiple government bodies, a landmark independent review, an NHS restructuring, an active court case, and now a clinical trial pause — all spread across half a decade. The terminology shifts. The institutions change. No single keyword follows it from start to finish.
At a Glance
- Latest development (February 20, 2026): The DHSC announced a pause after MHRA safety concerns and a recommendation for a minimum participant age of 14.
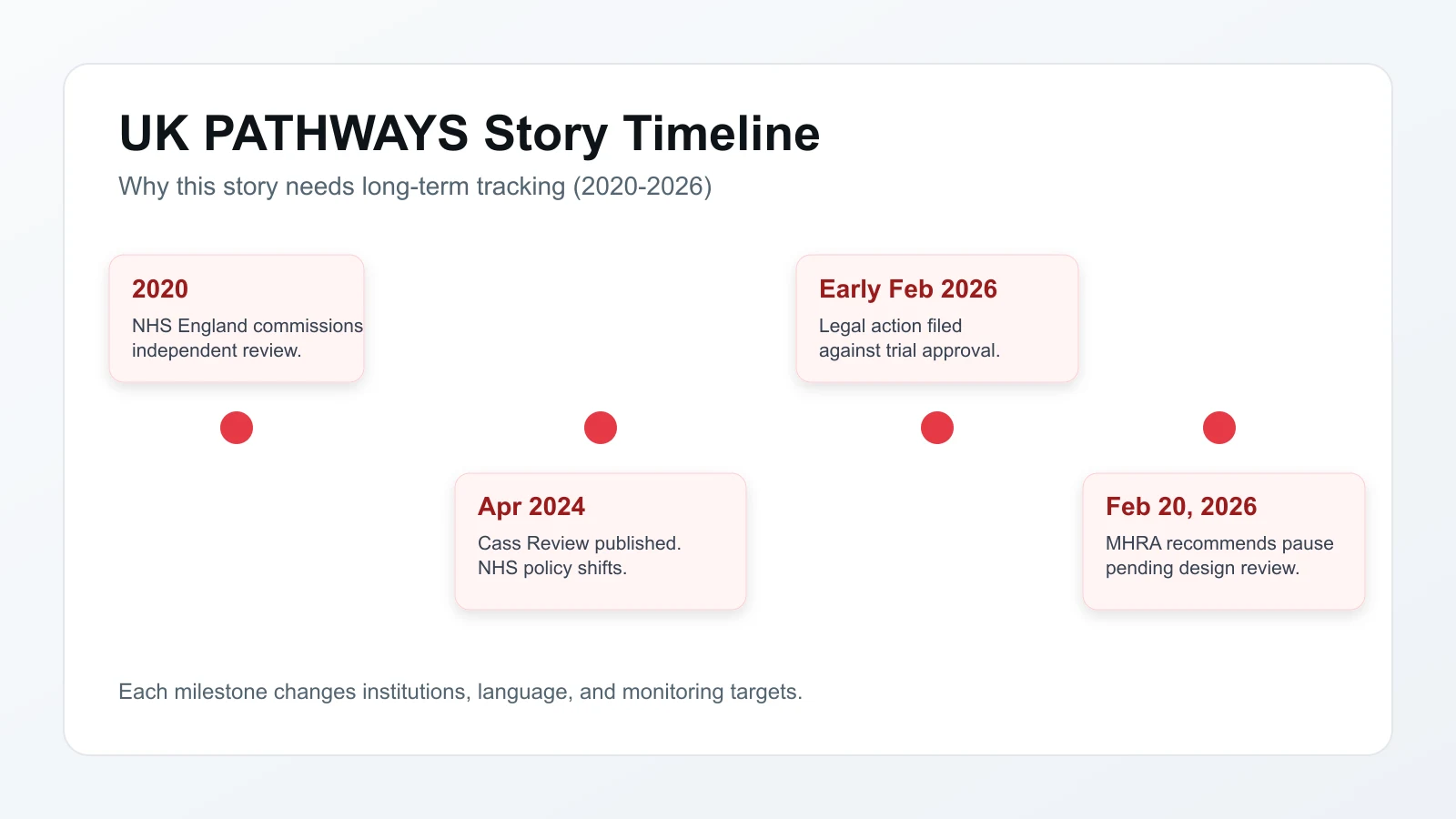
- How we got here: 2020 independent review commission → April 2024 Cass Review → early February 2026 legal challenge → February 2026 MHRA pause.
- What matters now: protocol changes, court outcomes, and the government's position can each move this story independently.
Timeline of key events from the 2020 review commission to the February 20, 2026 MHRA pause.
Why the UK Puberty Blocker Trial Is Hard to Follow
The PATHWAYS trial didn't appear from nowhere. It's the product of converging policy decisions, each with its own timeline and institutions.
The story starts with the Tavistock and Portman NHS Trust's Gender Identity Development Service (GIDS). For years, GIDS was the UK's only NHS gender clinic for young people — prescribing puberty-suppressing hormones to hundreds of patients annually. In 2020, NHS England commissioned an independent review of gender identity services for children. That review took four years.
In April 2024, Dr. Hilary Cass published her final report. She found the evidence base for puberty blockers was "remarkably weak" and that it was not possible to draw conclusions about long-term effects on development. The NHS then suspended routine prescriptions. GIDS was closed and replaced by a network of regional centres. The Cass Review did not call for a permanent ban; it called for formal research through properly designed clinical trials.
That became the PATHWAYS study, sponsored by King's College London and ethically approved by the Health Research Authority.
Then the legal challenges arrived. In early February 2026, campaigners including Keira Bell — whose earlier High Court case against GIDS had reshaped NHS prescribing policy — launched legal action against the Health Research Authority and the DHSC. They argued the ethical approval process was flawed and that the trial posed risks of irreversible harm.
Days later, the MHRA announced the pause.
Tracking this story requires watching four different entities across five years: Tavistock, the Cass Review, King's College London, the MHRA, plus the courts. Most keyword monitoring breaks down when the first institution hands the story to the next. Pingmer's thread on the trial tracks it as a single evolving narrative — one timeline from 2020 through the pause.
Institution handoff map showing how responsibility moved across services, reviewers, trial sponsors, and regulators.
Track this story on Pingmer — it's free →
What the MHRA Pause Actually Means
"Paused" is not the same as "cancelled." The MHRA's concerns relate to participant wellbeing and the proposed minimum age for trial participants. The regulator has not concluded the trial should never proceed — only that the current design needs review.
Discussions between the MHRA and King's College London are scheduled to begin following the announcement. Recruitment will not start until those issues are resolved.
Three things to watch next:
- The MHRA-KCL discussions — whether protocol amendments can address the age concern, and what timeline the regulator expects.
- The legal action — the judicial review case is still active. A successful challenge could halt the trial independently of the MHRA's concerns.
- Government position — the Department of Health's response if both tracks proceed simultaneously.
What to monitor next: protocol discussions, judicial review progress, and government positioning.
This story is unlikely to resolve quickly. The original Cass Review took four years. Clinical trial protocol amendments can take months. Legal proceedings move on their own schedule.
What a Story Like This Actually Needs
Multi-year policy stories are where conventional keyword tracking fails completely.
Say you set up keyword monitoring for "puberty blockers UK" in 2020. Here's what you'd have missed:
- 2022: Early design work on what would become PATHWAYS
- April 2024: Cass Review publication and immediate NHS policy change — the biggest single shift in the story
- Early 2026: Legal action filed against the ethical review process
- February 2026: MHRA pause and recruitment halt
The language around the story changed at every step. "Tavistock" became "GIDS" became "PATHWAYS." "Puberty blockers" appears in some sources as "puberty-suppressing hormones" or "GnRH analogues." A keyword match on any single term misses large chunks of the story's arc.
What works better: treating the story as a narrative rather than a search term. Following what's happening to the specific thread of events — the NHS's approach to gender medicine for young people — rather than keyword matches in articles that touch on it.
This is the same challenge that comes up with long-running regulatory stories in the US and with court cases that span years of filings and hearings. The institutions shift. The names change. The story continues.
How to Follow Health Policy Stories That Evolve Over Years
Whether you're following the PATHWAYS trial or any other complex health or regulatory story, a few principles matter.
Start with primary sources, not just coverage. For the PATHWAYS trial, primary sources include GOV.UK (DHSC announcements), MHRA statements, and Health Research Authority ethics decisions. These often precede or diverge from media coverage, sometimes by days.
Identify the relevant institutions up front. Write them down. For this story: MHRA, DHSC, King's College London, NHS England, and the courts. Monitor for each institution's involvement, not just a single keyword. When one institution hands the story to another, you follow it.
Watch for protocol shifts, not just outcomes. The meaningful developments in a clinical trial are rarely just "approved" or "cancelled." They're age limit changes, ethics board decisions, interim findings, and protocol amendments. These often skip the front page but shape what happens next.
Use something built for persistence. Most story monitoring tools optimize for recency — what happened in the last 24 hours. For stories like PATHWAYS, you need something that remembers eighteen months of context and surfaces a February 2026 update in the context of what happened in April 2024. That context is the difference between understanding the story and just seeing a headline.
Pingmer keeps a live, sourced timeline for the PATHWAYS trial, going back to when the story began.
For a broader look at what this kind of story tracking is and who uses it, see what story tracking means.
Start tracking the puberty blocker trial →
Frequently Asked Questions
What is the PATHWAYS trial?
PATHWAYS is a clinical research study sponsored by King's College London into the effects of puberty-suppressing hormones on young people experiencing gender incongruence. It was commissioned following the 2024 Cass Review, which found the existing evidence base for puberty blockers insufficient to guide clinical practice. The trial was designed to generate the controlled evidence the NHS needs to make future policy decisions.
Why was the PATHWAYS trial paused?
The MHRA raised "new concerns" about participant wellbeing in February 2026, recommending a minimum participant age of 14 due to "unquantified risk" of long-term biological harms. The Department of Health and Social Care announced the pause pending discussions between the MHRA and trial sponsor King's College London. Separately, active legal action from campaigners including Keira Bell is contesting the trial's ethical approval.
What was the Cass Review?
The Cass Review was an independent review of NHS gender identity services for children and young people, led by Dr. Hilary Cass and published in April 2024. It found the evidence for puberty blockers in gender medicine "remarkably weak" and called for a more cautious approach — including formal clinical research to generate the evidence base the field lacked. Its recommendations led to the NHS suspending routine puberty blocker prescriptions and commissioning the PATHWAYS study as the path forward.
How can I stay updated on the PATHWAYS trial?
Pingmer has a live thread tracking the full history of the story since 2020 — with sourced updates and notifications when something significant changes. See the live thread card above. Free signup, no commitment.